Formulating Flexibility with ActiMask® Taste Masking
Wayne Camarco, Global Head of Technical Development, and Donald Loveday, Ph.D., Global Business Development Manager, SPI Pharma
While drug discovery continues to accelerate, and the range of active pharmaceutical ingredients (APIs) available to patients continues to expand, most APIs are not palatable in oral solid dosage forms. Patient-centric formulations that mask the taste of APIs are a key factor in ensuring adherence to medication regimens and improving patient experiences. Wayne Camarco and Donald Loveday discuss SPI Pharma’s innovative technology to encapsulate API particles, the company’s relaunched Actimask® taste-masked ibuprofen product, and how their taste-masking formulation development services support product launches worldwide.
The Value of Patient-Centric Formulations
Patient non-compliance with treatment regimens costs the U.S. healthcare system billions of dollars each year. Many of the causes of poor medication adherence relate directly to properties of the drugs on the market themselves. Among oral solid dosage forms, many are large and difficult to swallow or present organoleptic issues like poor taste or mouthfeel. Some require frequent dosing that can be inconvenient, especially if they are not easy to take when on the go. These administration challenges are magnified among certain populations, including children, the elderly, and anyone with physiological difficulties in swallowing, making non-adherence an even more pressing issue for these patients.
Cap Gemini Consulting estimated 10 years ago that increasing adherence rates by only 10 percentage points would translate into a $41 billion pharmaceutical revenue opportunity in the United States and $124 billion globally. As importantly, those gains would be accompanied by improved health outcomes and decreased healthcare spending.1
The Importance of Taste Masking
The success of orally administered tablets that have residence times in the oral cavity depends on how easy and pleasant it is for patients to take them. Palatability, including mouthfeel (smooth vs. gritty) and taste, is therefore a very important consideration. Most active pharmaceutical ingredients (APIs), however, have a very bitter taste. Others have unpleasant odors or textures, and some may even be irritating. Thus, patient-centric formulations that can contribute to increased patient adherence must incorporate technology to not only overcome these negative attributes but create a pleasant taste and mouthfeel.
If a coating technology is to be employed, the coating must provide sufficient barrier properties to mask taste when the dose form is in the oral cavity yet dissolve quickly enough to not retard drug release when the drug particles pass into the GI tract. The next challenge is to formulate the taste-masked drug into a solid dosage form with acceptable organoleptic properties.
Taste-Masking Techniques
A range of taste-masking technologies are employed in the formulation of oral dosage forms. The simplest are flavorants and sweeteners. However, the use of these excipients is often insufficient to provide the right taste and texture to a given formulation. Changing the structure of the API so that it cannot interact with taste receptors is another approach. Salt formation and the generation of a pro-drug that is degraded in the gastrointestinal (GI) tract to release the actual API are two methods frequently used. Complexation with cyclodextrin or ion-exchange resins can also prevent interaction of the API with taste receptors. Spray granulation to form solid dispersions and co-extrusion are other options.
Applying a barrier coating to API particles or the final tablet is another approach. Synthetic polymer coatings can be applied using various well-established coating techniques, such as fluid-bed and Wurster/rotor coating. Coacervation — separating a solution into two immiscible liquid phases — is also applicable for producing thin protective barriers that aid in taste masking
Many Challenges to Effective Taste
- Bitterness
- Dose
- Particle size
- Particle shape
- Solubility
If a coating technology is to be employed, the coating must provide sufficient barrier properties to mask taste when the dose form is in the oral cavity yet dissolve quickly enough to not retard drug release when the drug particles pass into the GI tract. The next challenge is to formulate the taste-masked drug into a solid dosage form with acceptable organoleptic properties. Finding the balance between dissolution and taste masking while achieving desirable mouthfeel is not easy and requires materials and formulation expertise.
Actimask® Technology: A Better Barrier Solution
SPI Pharma has developed a taste-masking technology that involves the production of a thin (micron), uniform, smooth hydrogel coating encapsulating API particles (see Figure 1). The proprietary Actimask® aqueous coacervation process uses gelatin as the cationic, hydrophilic polymer. By carefully controlling the pH and temperature and through the use of specific initiating agents, the gelatin is forced out of solution to form a thin layer on the API particles.
Figure 1:
API particle encapsulated in Actimask® barrier coating
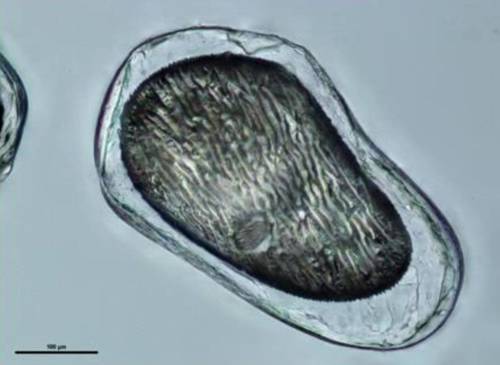
Because the Actimask® process is water-based, there is no concern of residual solvents, as can be the case with many other taste-masked products. When APIs are insoluble or only modestly soluble in water, high assays ( >85%) are readily obtained, allowing for the formulation of smaller tablets than can be realized using other coating technologies. The thinness of the coating layer also does not impact the particle size, and thus the particle size of the coated particles is controlled by the API particle size.
Notably, Actimask® technology provides an excellent taste barrier and mouthfeel without hindering API release and onset of therapeutic relief. The hydrogel layer is smooth and slippery, providing excellent flowability for processing and formulation, as well as an attractive mouthfeel and easy swallowing for a pleasant patient experience. The coating is also robust and remains intact during direct compression of the API particles into traditional tablets, orally disintegrating tablets (ODTs), or orally dispersible powders (ODPs). Finally, Actimask® coatings meet the requirements of various monographs for dissolution and other properties.
SPI Pharma offers two Actimask® products: taste-masked acetaminophen and ibuprofen, two popular analgesic/antipyretic drugs that have historically been challenging to formulate given their bitterness and other properties.
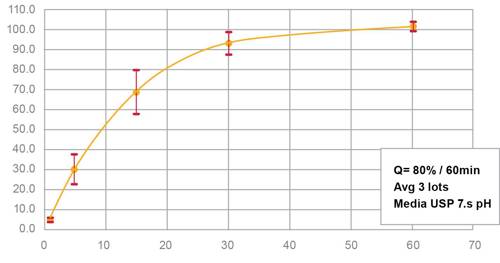
Figure 2:
In vitro dissolution of Actimask® IBU with 86% assay
Relaunch of Actimask® Ibuprofen
It is well known that uncoated ibuprofen causes an intense burning sensation when exposed to the oral cavity and throat. Actimask® taste-masked ibuprofen is an ideal choice for the formulation of chewable or orally dispersible dosage forms, because the Actimask® coating reduces — to extremely low levels — the burning sensation that many patients experience with poorly masked or unmasked ibuprofen products.
Actimask® Ibuprofen exhibits minimal drug release in the oral cavity during the first few minutes of exposure, thus avoiding the unpleasant taste and sensation associated with this API when swallowed. However, at gastric pH, it exceeds USP/EP compendial dissolution requirements (see Figure 2). An in vivo pharmacokinetic study has shown that formulations with Actimask® Ibuprofen are comparable to other marketed products (see Figure 3).
Figure 3:
In vivo dissolution of Actimask® IBU with 86% assay in ODT and ODP formulations
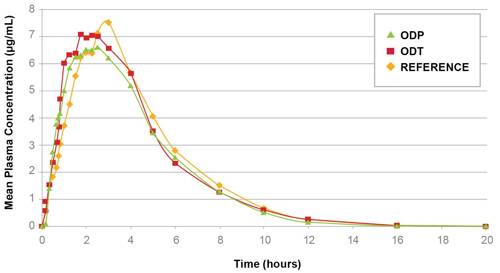
Randomized, open-label, crossover design in 13 adults using 100 mg ODT and ODP formulations containing Actimask® Ibuprofen 92S (92% assay) vs. 100 mg Junior Advil® Chewable
The aqueous hydrophilic coating delivers an excellent taste barrier without any concern regarding residual solvents. In addition, the uniform hydrogel coating provides a smooth surface that has an excellent mouthfeel and is easy to swallow. Furthermore, the high assay content of Actimask® Ibuprofen allows for formulation flexibility and also helps to reduce the tablet weight of high-dose formulations.
To demonstrate the flexibility of Actimask® Ibuprofen, four different formulations were prepared using various SPI Pharma excipients and technologies: an ODT, chewables, and a fast-melt product. The formulations each contained an orange flavorant and sucralose sweetener additives, as well as one or more of SPI’s proprietary excipients, including PharmaBurst® 500 (ODT), Mannogem® XL Opal (fast-melt and chewable), and Mannogem® XL Ruby (chewable) excipients. In all cases, the tablet thickness, hardness, disintegration time, and dissolution profiles all met expectations.
Application-First Excipient Company
SPI Pharma consistently focuses on product innovation with the goal of solving customer challenges. Combining our formulation experience with deep scientific knowledge of customer end uses is a fundamental element of the SPI Pharma brand. We do not seek applications for our excipients, but instead design our excipients to address specific application needs.
This approach has been clearly demonstrated across our history. PharmaBurst® was the first ODT excipient platform developed for use in directly compressible ODT products. Seventeen years later, we introduced UltraBurst®, which offers an improved dissolution time. CS-90 and CM-90 are unique functional calcium excipients for use in antacid and nutritional tablets. The Mannogem® range of mannitol excipients offer innovative solutions for various oral solid dosage forms. Actimask® technology is yet another example.
Offering More than Just Excipients
Our range of taste-masking formulation development services includes the incorporation of our highly functional excipients along with our co-processed delivery systems. In addition to coacervation, SPI Pharma has the ability to develop taste-masked formulations leveraging complexation with ion-exchange resins, traditional polymer coating processes, and powder coating for water-soluble APIs.
These services are accessed through our Pharmasolutions offering, which is a comprehensive drug development service focused on patient-friendly dosage forms that can deliver convenience, compliance, and efficacy. Unlike a contract research organization, SPI Pharma is an ingredients manufacturer with materials and functionality expertise. We can use that deep material and formulation expertise to help bring new customer products to market quickly and to expand the life cycle of existing products by leveraging unique, patient-centric dosage forms and comprehensive regulatory capabilities.
To date, we have supported more than 60 global product launches. Our applied innovation group can provide assistance with technology and business development, licensing, formulation development and optimization, product prototyping, dossier development, stability testing, analytical method development and validation, and technology transfer.
ABOUT SPI PHARMA
SPI Pharma provides formulation innovation and support to pharmaceutical clients in more than 55 countries. Its products include antacid actives, excipients, taste-masking and fast-dissolve technologies, drug delivery systems for tablets, and a variety of other innovations for patient-friendly dosage forms. The company’s functional excipients can be used in a wide range of customer applications, including soft chew, chewable, swallow tablets, lozenges, orally disintegrating, and soft gel dosage forms. SPI Pharma is headquartered in Wilmington, Delaware, and has locations across the United States, France, and India.
References
1. Forissier, T. and K. Firlike. Estimated Pharmaceutical Revenue Loss Due to Medication Non-Adherence. Cap Gemini Consulting. 2012.

